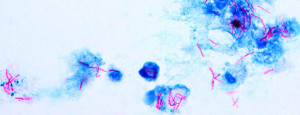
ACID FAST STAINING
Certain bacterial species have unusual lipids (mycolic acid) in their cell walls. Mycolic acid renders the cell wall very waxy and impenetrable by aqueous or ordinary aniline dyes.
In 1882 Robert Koch reported the discovery of the tubercle bacillus and described the appearance of the bacilli resulting from a complex staining procedure. Koch and Ehrlich simultaneously introduced a method for staining the previously undetectable Mycobacterium tuberculosis. Modifications by Ziehl and Neelsen produced the commonly used carbol-fuchsin solution which requires steaming to drive the stain in. (The melting point for mycolic acid is 56oC).
Muller and Chermock modified carbol-fuchsin for use at room temperature by addition of a surfactant (wetting agent). Acid-alcohol removes stain from most cellular and tissue elements. The mycolic acid, however, resists penetration by this differentiating agent, leaving acid-fast bacteria red against a colorless background.
Acid fast bacilli resists decolourization with acid alcohol & retains primary stain i.e., carbol fuchsin.
Either methylene blue or malachite green is used as a counter stain to aid in the localization of cellular material on the specimen. The acid fast stain is used routinely on sputum samples for preliminary diagnosis of active tuberculosis.
The Ziehl-Neelsen method has endured as a reliable and effective way to demonstrate the acid fast bacteria. In this method heat is used to help drive the primary stain into the waxy cell walls of these difficult-to-stain cells. The use of heat in this method has been the reason that this technique is called the “hot staining” method.
In 1915, Kinyoun published a method that has become known as the “cold staining” method because the heating step was removed in favor of using a higher concentration of the carbol fuchsin primary stain.A. Ziehl-Neelsen method for acid-fast staining
Carbol fuchsin stain:
Basic fuchsin, 0.3 g
Ethanol, 95% (vol/vol), 10 ml
Phenol, heat-melted crystals, 5 ml
Distilled water, 95 ml. Dissolve the basic fuchsin in the ethanol; then add the phenol dissolved in the water.
Mix and let stand for several days. Filter before use.
Decolorizing solvent:
20% Sulphuric acid solution: Place 800ml water in a large flask .
Add 200ml concentrated sulphuric acid (about 98%) . The acid should be poured slowly down the side of the flask into the water about 50 ml at a time. The mixture will become hot . Mix gently, add remainder of the acid in the same manner.
Note: The acid must be added to the water.It is dangerous to add the water to the acid. Great care is required.
Alcohol 95%:
Ethanol 95 ml plus water to 100ml
OR
Ethanol, 95% (vol/vol), 97 ml Hydrochloric acid (concentrated), 3 ml
Counterstain:
Methylene blue chloride, 0.3 g
Distilled water, 100 ml
B. Kinyoun method for acid-fast staining
Kinyoun carbol fuchsin solution:
Solution A. Dissolve 4 g of basic fuchsin in 20 ml of ethyl alcohol.
Solution B. Dissolve 8 g of phenol (melted) in 100 ml of distilled water.
Mix solutions A and B together and allow to stand for a few days.
Acid-alcohol decolorizing agent:
Ethanol, 95% (vol/vol), 97 ml
Hydrochloric acid (concentrated), 3 ml
Methylene blue counterstain:
Methylene blue chloride, 0.3 g
Distilled water, 100 ml , Dissolve by shaking.
A. Ziehl-Neelsen method for acid-fast staining:
- Films are made dried and fixed by flaming.
- Cover the slide with filtered carbol fuchsin and heat until steam rises. Allow the preparation to stain for 5 min, heat gently .The stain must not be allowed to evaporate and dry on the slide. If necessary pour on more carbol fuchsin to keep the whole slide covered.
(The slide may be heated with a torch prepared by twisting a small piece of cotton wool on to the tip of an inoculating wire and soaking it in methylated spirit before lighting. when steam rises from the slide, remove and extinguish the torch. after about 1 minute recharge the torch with spirit, relight it and again heat the slide until the steam rises, continue this way for 5 min.)
- Wash with water.
- Cover the slide with 20% sulphuric acid. Keep for 1` min. wash with water.
- Treat with 95% alcohol for 2 min
- Wash with water
- Counterstained with Loeffler’s methylene blue. Or dilute malachite green for 15-20 sec.
- Wash, blot dry and mount.
B.Kinyoun method for acid-fast staining:
1. Heat fix an air dried smear at 80oC for at least 15 minutes or for 2 hours on an electric hot plate at 65oC – 70oC
2. Flood slides with Kinyoun’s carbol fuchsin reagent and allow to stain for 5 minutes at room temperature.
3. Rinse with deionized water and tilt slide to drain.
4. Decolorize with acid-alcohol for 3 minutes and rinse again with deionized water.
5. Redecolorize with acid-alcohol for 1-2 minutes or until no more red color runs from the smear.
6. Rinse with deionized water and drain standing water from the slide surface by tipping the slide.
7. Flood slide with methylene blue counterstain and allow to stain for 4 minutes.
8.Rinse with distilled water and allow to air dry.
9. Examine under high dry (400X) magnification, and confirm acid-fast structures under oil immersion (1000X).
Interpretation
Acid-fast bacilli take pink red colour against blue background.
Exercise
Q1. Draw well labelled diagram of cell wall of acid fast bacteria. Q2. Enlist the names of acid fast bacteria.
Reference
A.V. Bhonsle & A.G. Karpe, 2007, General Microbiology Laboratory Manual
Cruickshank, Medical Microbiology
http://www.microbelibrary.org/component/resource/laboratory-test/2870-acid-fast-stain-protocols
Kinyoun, J. J. 1915. A note on Uhlenhuth’s method for sputum examination for tubercle bacilli. Am. J. Public Health 5:867.